
😂 Which is an organic molecule. CHEMISTRY II WATER AND ORGANIC
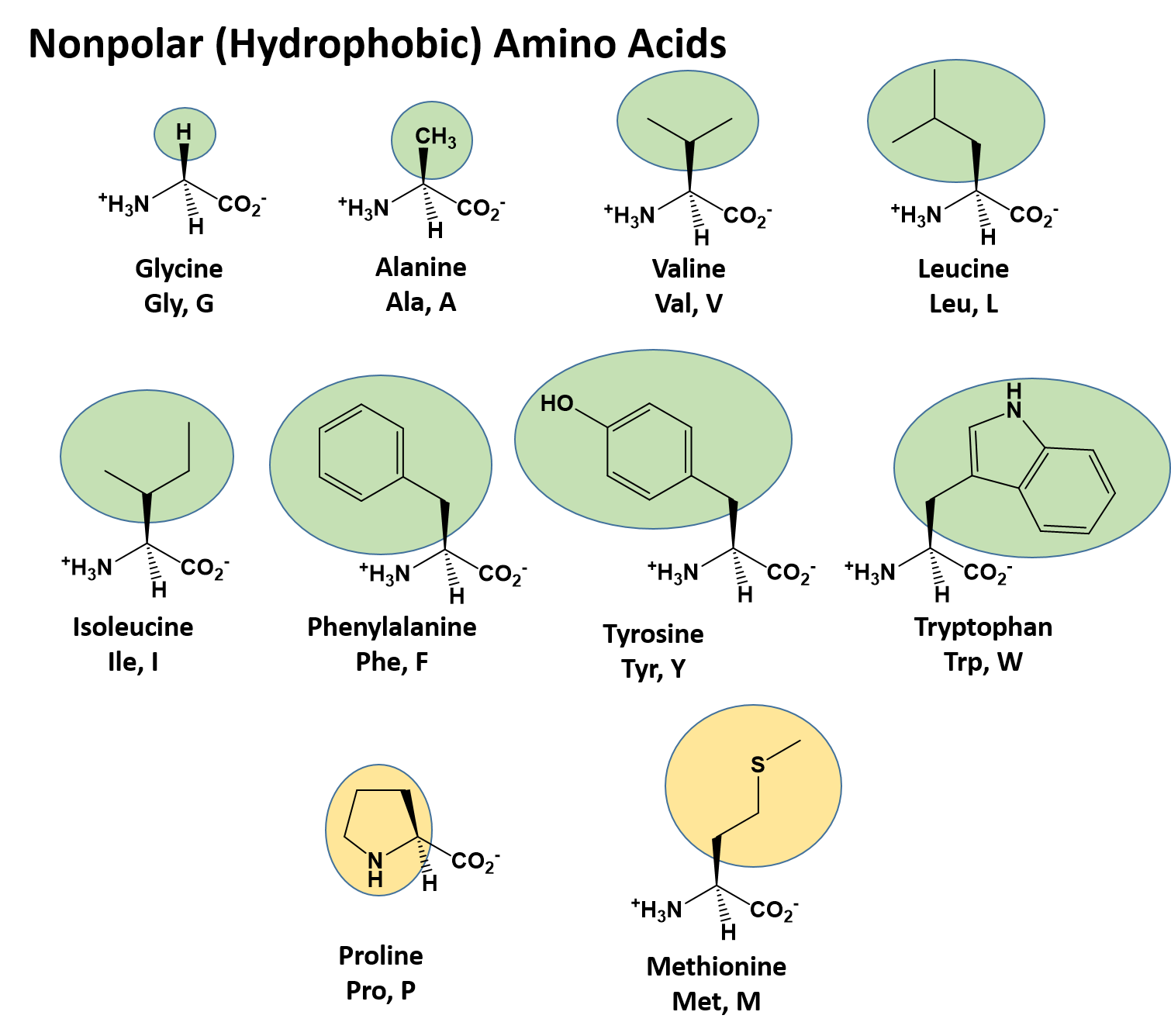
The structures, abbreviations (both three- and one-letter), and pK a values of the 20 amino acids commonly found in proteins are shown in Table 26.1.All are α-amino acids, meaning that the amino group in each is a substituent on the α carbon—the one next to the carbonyl group. Nineteen of the twenty amino acids are primary amines, RNH 2, and differ only in the nature of their side chain.
CH150 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
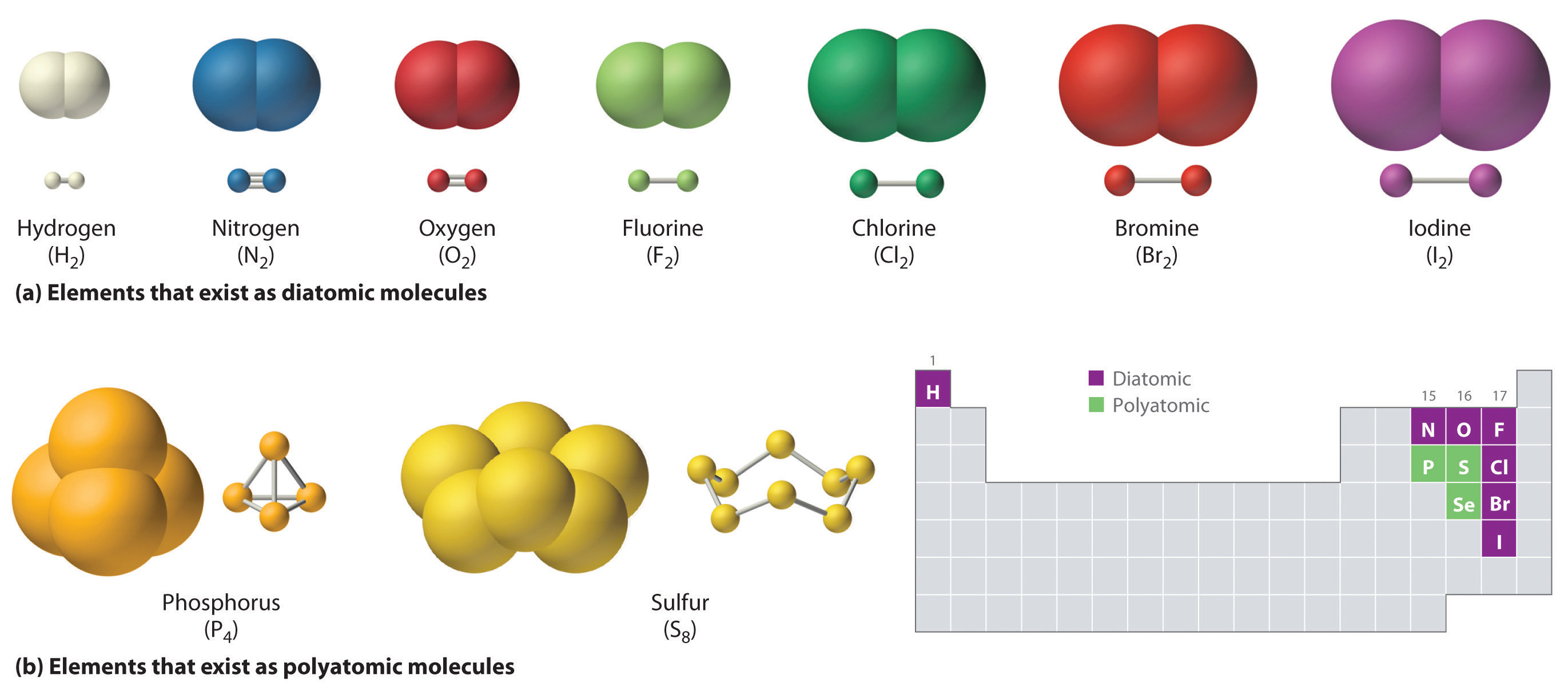
The chemistry of these compounds is called organic chemistry. Hydrocarbons are organic compounds composed of only carbon and hydrogen. The alkanes are saturated hydrocarbons—that is, hydrocarbons that contain only single bonds. Alkenes contain one or more carbon-carbon double bonds. Alkynes contain one or more carbon-carbon triple bonds.

Organic Chemistry 101 Nomenclature Organic chemistry reactions
Figure 26.1. 2: The Tetrahedral Methane Molecule. Methane (CH 4 ), ethane (C 2 H 6 ), and propane (C 3 H 8) are the beginning of a series of compounds in which any two members in a sequence differ by one carbon atom and two hydrogen atoms—namely, a CH 2 unit. The first 10 members of this series are given in Table 26.1.
Organic Molecules Mrs. CovarrubiasAdvanced Biology
Chapt01+Fig+Cont+ZCD1. 1: Organic Molecules and Chemical Bonding. Preview 1-3. 1.1 Organic Molecules 1-4. Bonding Characteristics of Atoms. (1.1A) 1-4. Bonds and Unshared Electron Pairs for C, N, O, and F. Bonds and Unshared Electron Pairs for Other Atoms. Structures of Organic Molecules.
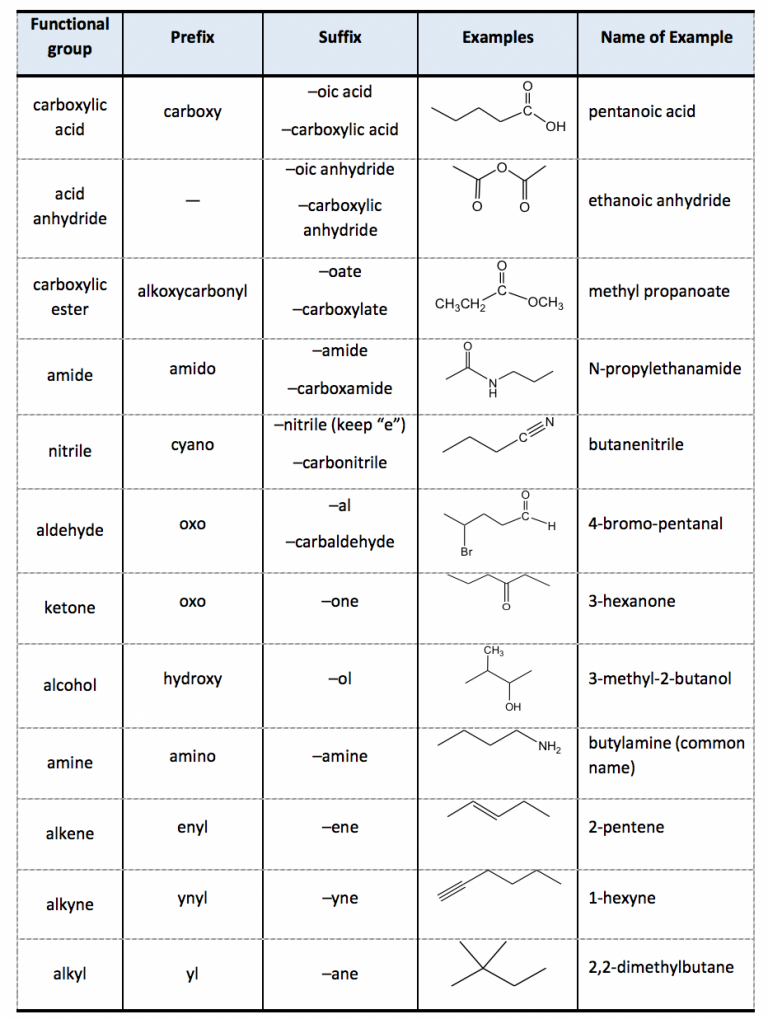
Functional Groups in Organic Compounds
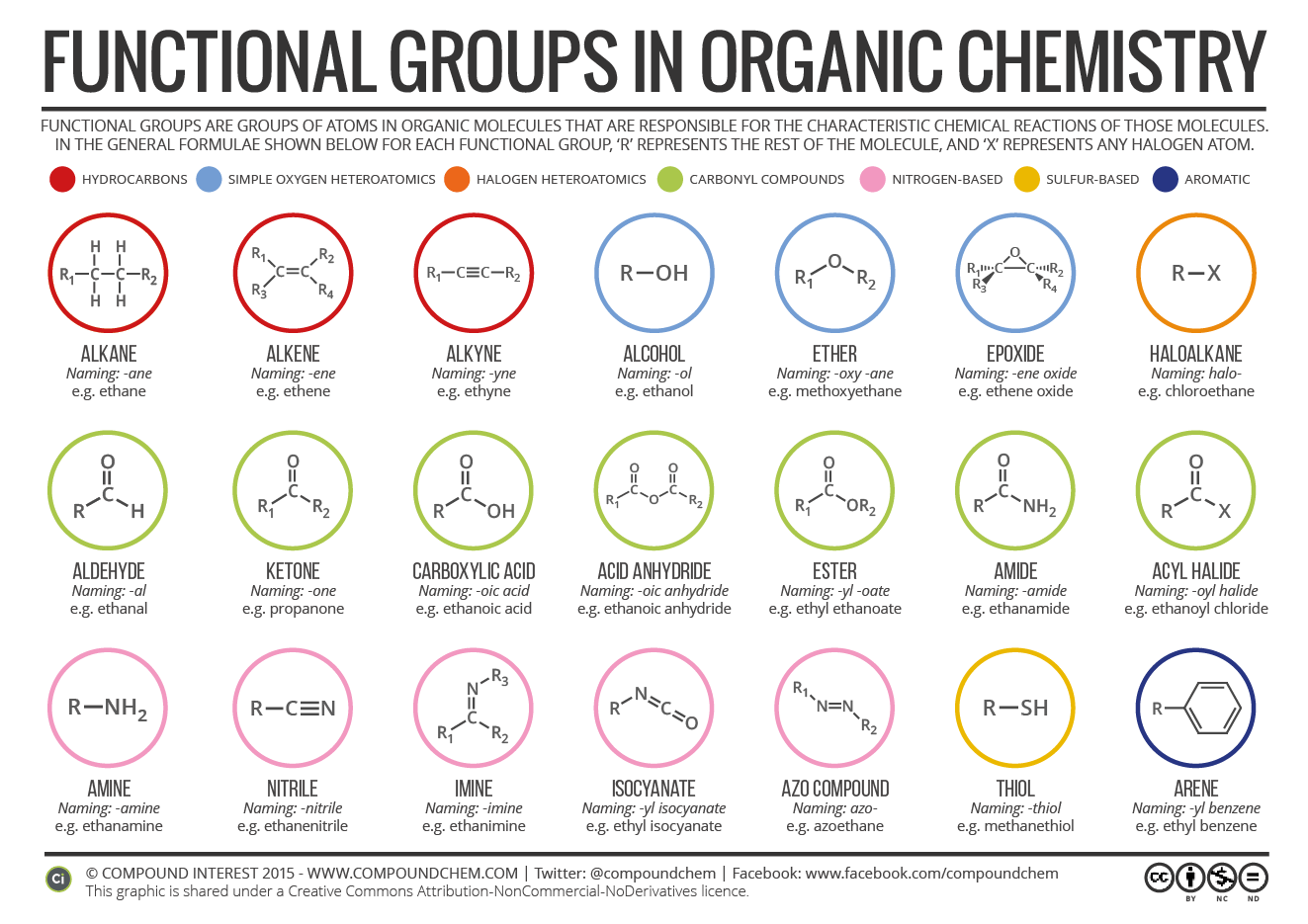
Substitutive nomenclature is the main method for naming organic-chemical compounds. It is used mainly for compounds of carbon and elements of Groups 13-17. For naming purposes, a chemical compound is treated as a combination of a parent compound (Section 5) and characteristic (functional) groups, one of which is
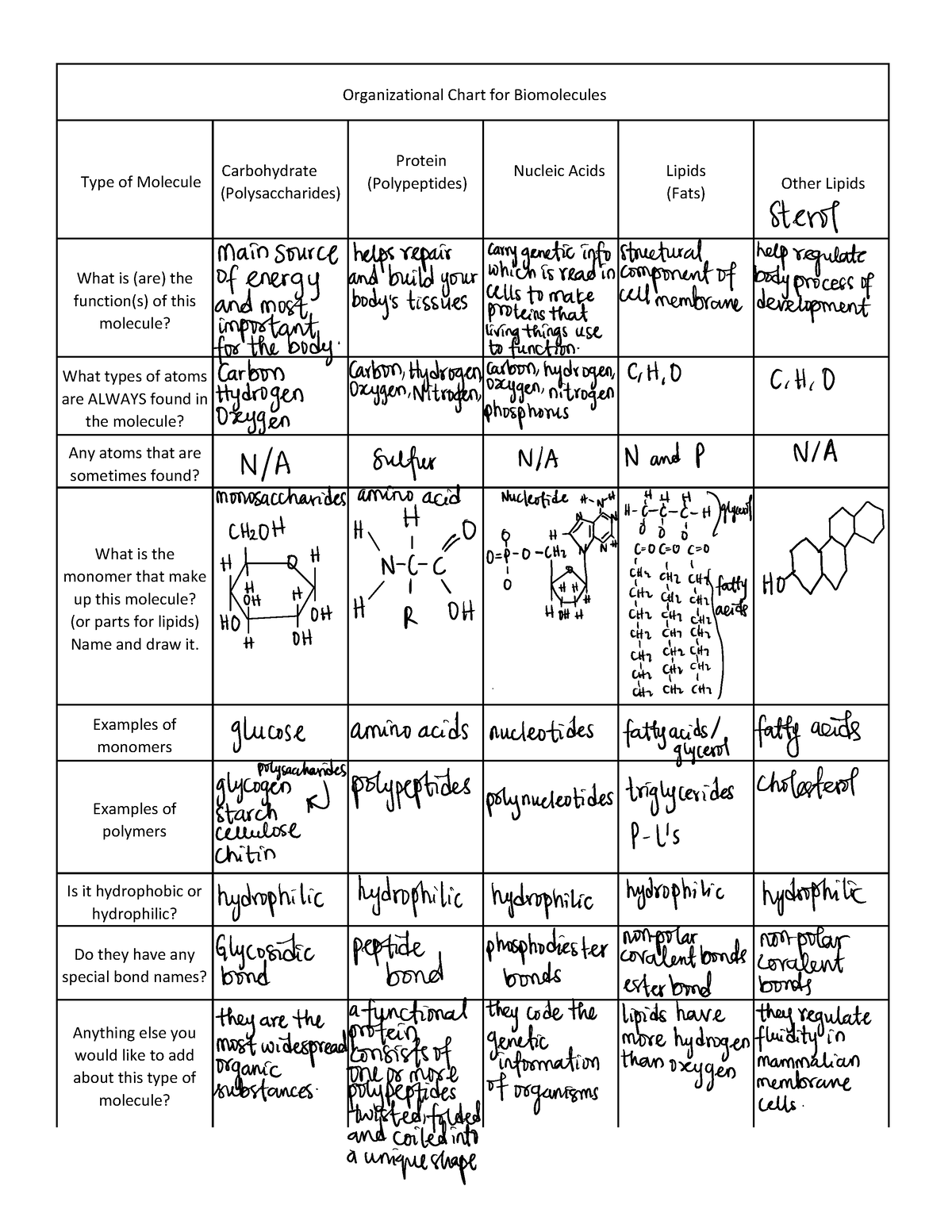
Organic Molecules worksheet Organizational Chart for Biomolecules
For many purposes, ball-and-stick models of organic compounds give useful information about the spatial relationships of the atoms, and for \(CX_4\) the angles between sticks are set at \(109.5^\text{o}\) (Figure 2-1). Organic molecules strongly resist deformation forces that alter their valence angles from normal values.

Organic Molecules Chart
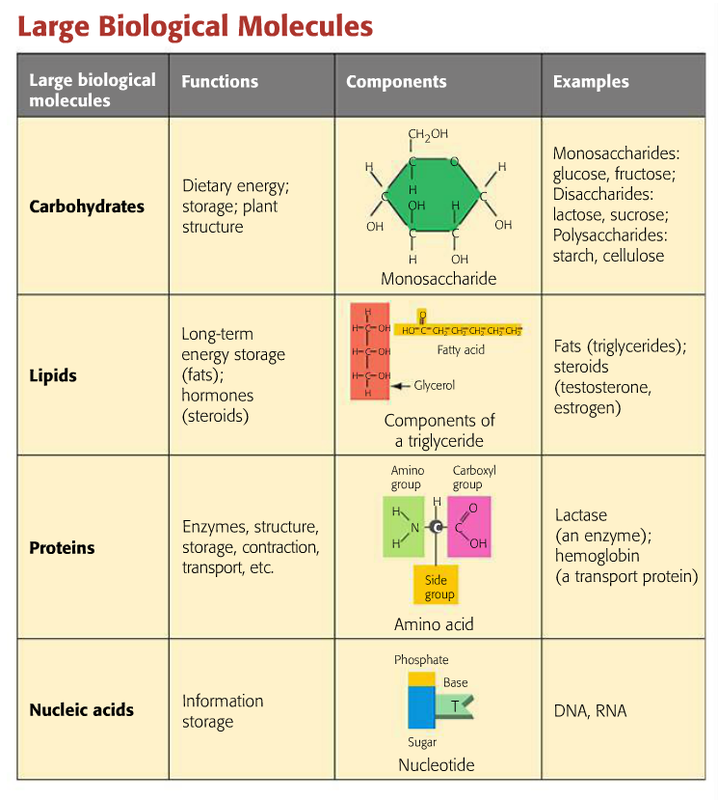
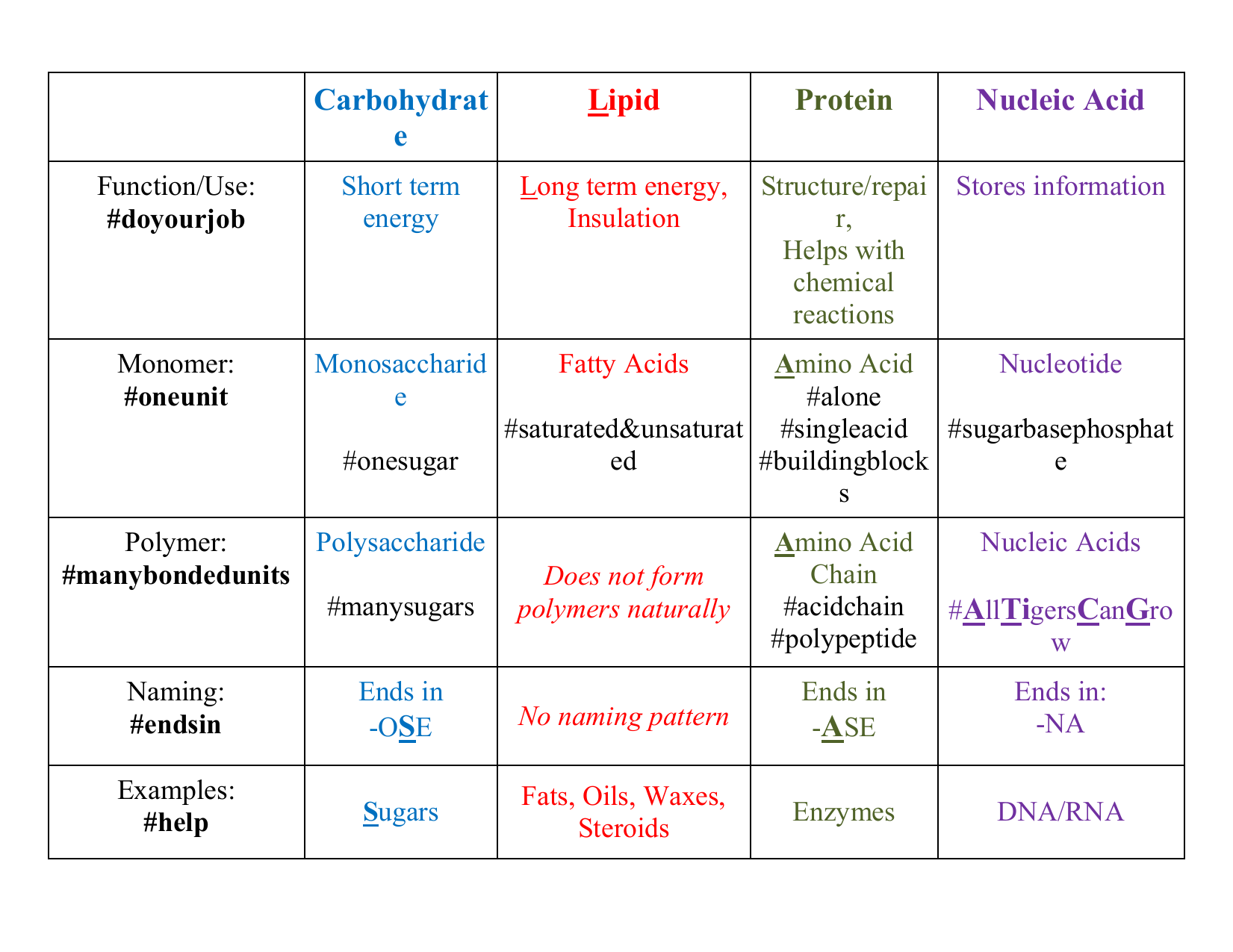
Carbohydrates are the first class of organic molecules. The simplest kind of carbohydrate includes monosaccharide simple sugars, which have a basic formula: two hydrogens and one oxygen for every carbon atom or one water for every carbon. Glucose is a common carbohydrate whose formula is C6H12O6. Sugars can be either a single sugar molecule to.

Probable chemical mechanisms of different classes of organic compounds
Organic chemistry 14 units. Unit 1 Structure and bonding. Unit 2 Resonance and acid-base chemistry. Unit 3 Alkanes, cycloalkanes, and functional groups. Unit 4 Stereochemistry. Unit 5 Substitution and elimination reactions. Unit 6 Alkenes and alkynes. Unit 7 Alcohols, ethers, epoxides, sulfides. Unit 8 Conjugated systems and pericyclic reactions.
Organic Molecules Chart
They are small, simple compounds that play important roles in the cell, although they do not form cell structures. Most of the carbon found in organic molecules originates from inorganic carbon sources such as carbon dioxide captured via carbon fixation by microorganisms. Exercise 7.1.2 7.1. 2. Describe the most abundant elements in nature.
Organic Molecule Chart
A further complication is that, even outside of a biological context, many simple organic molecules are known almost universally by their 'common', rather than IUPAC names. The compounds acetic acid, chloroform, and acetone are only a few examples. In biochemistry, nonsystematic names (like 'cocaine', 'capsaicin', 'pyruvate' or.

PPT Organic Molecules and Carbohydrates PowerPoint Presentation, free
1. Organic compounds containing substituents from Group C are named following this sequence of steps, as indicated on the examples below: •Step 1. Find the longest continuous carbon chain. Determine the root name for this parent chain. In cyclic compounds, the ring is usually considered the parent chain, unless it is
CH103 Chapter 8 The Major Macromolecules Chemistry
Introduction. In its simplest definition, organic compounds include all molecules that contain carbon. By this definition, simple molecules such as carbon monoxide (CO) and carbon dioxide (CO2) would be defined as organic molecules, however, these simple molecules behave more like inorganic molecules than organic molecules.

Organic functional groups chart expanded edition M A N O X B L O G
The purpose of this chart will be clear if you've got a background in chemistry. If you haven't, it's a useful tool to decode the different parts that make up molecules in organic chemistry. All carbon-based (organic) molecules contain functional groups - some more than one of them - and they're what gives molecules their particular.

Chemical structure and common names of the 16 organic compounds used in
Table 2.4 Subordinate Groups. We will go through several examples for more details about the naming rules. 1. The parent structure is the 6-carbon carboxylic acid with a double bond, so the last name comes from "hexene". To add the suffix, the last letter "e" will be dropped, so the parent name is "hexeneoicacid".
3.2 Conformations of cyclic organic molecules Chemistry LibreTexts
Tim Soderberg. University of Minnesota Morris. An understanding of the various types of noncovalent forces allows us to explain, on a molecular level, many observable physical properties of organic compounds. In this section, we will concentrate on solubility (especially solubility in water), melting point, and boiling point.

2.3 Biologically Important Macromolecules Biology LibreTexts
Structure search. Search by Structure or Substructure. Upload a structure file or draw using a molecule editor.